How To Create Molecular Orbital Diagrams
11.5: molecular orbital theory Chemistry molecular orbital diagram heteronuclear molecule, png Molecular orbital theory
What are antibonding molecular orbitals? + Example
Orbital molecular bonding orbitals find determine electrons geometry level o2 molecules calculate unpaired covalent libretexts diamagnetic paramagnetic atoms chem Diagram orbital molecular ozone bonding orbitals antibonding mo theory molecule non electrons bonds delocalized chemistry resonance example multiple polyatomic pi Cl2 molecular orbital diagram
Orbital molecular diatomic molecules diagram chemistry theory orbitals diagrams energy bond bonding level libretexts cl2 delocalized second electron homonuclear row
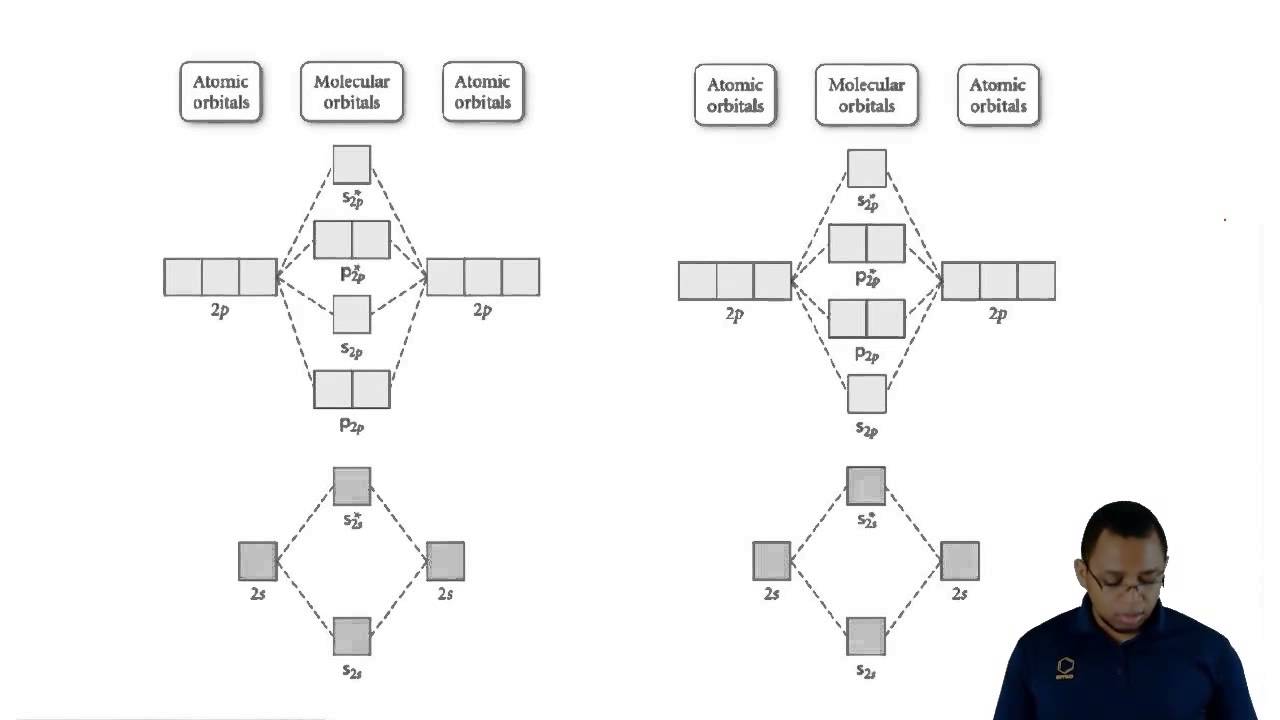
Heteronuclear molecular orbital diagram molecule chemistry atomicOrbital diagrams ion simplified lim atoms heteronuclear diatomic molecules Understanding molecular orbital theoryN2 2 molecular orbital diagram.
What are antibonding molecular orbitals? + exampleOrbital cl2 orbitals diatomic energy electron bonding delocalized molecules electrons valence diagrams libretexts paramagnetic chemistry heteronuclear 3s Electron orbitals electrons quantum chemistry numbers electronic structure introductory model orbital atoms figure atomic arrangement number energy ball libretexts chapterChapter 6.5 delocalized bonding and molecular orbitals.

Introductory chemistry 1.0
Molecular orbital diagrams simplifiedOrbital molecular theory Orbital molecular n2 orbitals bond diatomic practice atomic valence o2 c2 filled homonuclear majors libretexts sp3 molecule cnx chem ruleMolecular orbital orbitals sulfur bonding valence delocalized electrons libretexts chapter atom contributes atomic chem sponsored.
.